Ivan Stranski (Bulgarian: Иван Николов Странски; German: Iwan Nicolá Stranski; 2 January 1897 – 19 June 1979) was a Bulgarian physical chemist whose groundbreaking work laid the foundation for modern crystal growth theory. He is widely recognized as the founder of the Bulgarian school of physical chemistry and one of the most influential scientists in the field of surface science and materials physics.
Ivan Stranski was not only a pioneering scientist but also a central figure in the history of crystal growth science, shaping how modern researchers understand atomic structures and material formation. Known as the founder of crystal growth theory, Stranski’s work laid the foundation for breakthroughs such as the Stranski–Krastanov growth model and the Kossel–Stranski model explained in modern physics. As one of the most important figures in physical chemistry in Bulgaria, his contributions continue to influence today’s technologies, from semiconductors to nanotechnology. For anyone wondering who was Ivan Stranski, his legacy stands alongside the most famous Bulgarian chemists in scientific history.
His name lives on through two of the most important concepts in materials science:
These are not just historical theories—they are still used today in nanotechnology, semiconductors, quantum dots, and thin-film engineering.
Stranski’s life represents something bigger than science: it is the story of a Bulgarian scholar who built an entire scientific discipline from scratch and then helped rebuild European academia after World War II.
Early Life and Education
Ivan Stranski was born in Sofia, Bulgaria, at a time when the country’s scientific institutions were still developing. Bulgaria had only recently begun establishing modern universities, and opportunities for advanced scientific research were limited.
From an early age, Stranski showed exceptional intellectual ability. He pursued higher education during a period when physical chemistry was emerging as one of the most important scientific disciplines in the world. This field combined physics and chemistry to explain how matter behaves at the atomic and molecular levels.
Unlike many of his contemporaries, Stranski did not remain confined within national borders. He became part of a new generation of scientists who bridged Eastern and Western Europe, contributing to a shared scientific language that transcended politics.
Building Bulgarian Physical Chemistry

One of Ivan Stranski’s greatest achievements was not only theoretical—it was institutional. He did not simply contribute to science; he helped build an entire scientific discipline in Bulgaria from the ground up.
At Sofia University, Stranski founded and led the Department of Physical Chemistry, effectively establishing the field as an independent academic discipline in the country.
A scientific landscape in its infancy
At the time, Bulgaria faced significant challenges in developing advanced scientific research:
- Limited infrastructure for experimental chemistry
- Fragmented and loosely organized research efforts
- Lack of a unified national scientific school in physical chemistry
Scientific work existed, but it lacked coordination, depth, and international integration.
Transforming a system
Stranski fundamentally changed this landscape.
He introduced a structured, modern approach to scientific education and research, aligned with leading European standards. His efforts included:
- Creating formal academic programs in physical chemistry
- Developing research-focused curricula that emphasized both theory and experimentation
- Establishing laboratory practices based on contemporary scientific methods
This brought Bulgarian science closer to the level of major research centers in Germany and Western Europe.
Training a new generation
Perhaps his most lasting contribution was his role as a mentor and educator.
Stranski trained a generation of scientists who would go on to expand his work and contribute internationally. Among them was Lyubomir Krastanov, who later co-developed the Stranski–Krastanov growth model.
Through his students, Stranski’s influence extended far beyond his own research.
Introducing European scientific standards
Stranski served as a bridge between Bulgarian academia and the broader European scientific community.
He introduced:
- Rigorous theoretical frameworks
- Advanced experimental methodologies
- A research culture focused on innovation and publication
This integration ensured that Bulgarian science was no longer isolated but actively participating in global scientific progress.
A lasting institutional legacy
Because of these contributions, Ivan Stranski is widely regarded as:
👉 The architect of Bulgarian physical chemistry
He did not just advance knowledge—he created the conditions for knowledge to grow.
His work transformed Bulgaria from a country with limited scientific infrastructure into one capable of producing internationally recognized research in physical chemistry.
Because of this, he is considered:
👉 The architect of Bulgarian physical chemistry
His students, including future collaborators like Lyubomir Krastanov, continued his work and expanded his theories internationally.
👉 Stranski stands alongside other pioneers featured in Bulgarian scientists who changed the world
The Birth of Crystal Growth Theory

During the early 20th century, one of the biggest unanswered questions in science was:
👉 How do crystals actually form and grow?
Scientists had long observed crystals in nature—from minerals and metals to ice and salt—but the underlying mechanisms remained unclear. While their geometric shapes were well documented, the atomic processes responsible for their formation were not yet understood.
From observation to explanation
Before Ivan Stranski’s work, crystal growth was largely a descriptive field. Researchers could observe patterns, but they lacked a unified theory explaining how individual atoms organize into precise, repeating structures.
Stranski helped change that.
He introduced a powerful thermodynamic and kinetic framework that explained crystal growth at the atomic level. His approach combined:
- Thermodynamics – determining which structures are energetically stable
- Kinetics – explaining how fast and in what way atoms attach and rearrange
👉 To better understand the science behind crystal growth, see our guide on Bulgarian Scientific Discoveries that Changed the World.
What his theory explained
Stranski’s work provided answers to fundamental questions:
- How atoms attach to surfaces
Atoms do not stick randomly—they follow energy-driven pathways - How layers form during growth
Crystal surfaces develop through organized, stepwise layer formation - Why different growth patterns occur
Some materials grow smoothly, while others transition into island structures due to stress and energy conditions
This was a major breakthrough.
👉 Crystal growth was no longer mysterious—it became predictable and mathematically describable
From descriptive to predictive science
Stranski’s contributions transformed crystal growth into a true scientific discipline.
Instead of simply observing crystals, scientists could now:
- Predict how materials would grow
- Control surface structures
- Design materials with specific properties
This shift marked the birth of modern surface science and materials engineering.
Why it matters today
The principles Stranski helped establish are now essential to many of the technologies that define the modern world.
They are used in:
- Semiconductor chips – the core of computers and smartphones
- Solar panels – optimizing energy conversion efficiency
- Nanotechnology – building structures at the atomic scale
- Advanced materials engineering – designing stronger, lighter, and more efficient materials
Lasting significance
Stranski’s work answered a fundamental question about nature—but its impact extended far beyond theory.
👉 Understanding how crystals grow made it possible to engineer the materials that power modern technology
From microchips to renewable energy systems, his ideas continue to shape the future.
Stranski–Krastanov Growth Explained
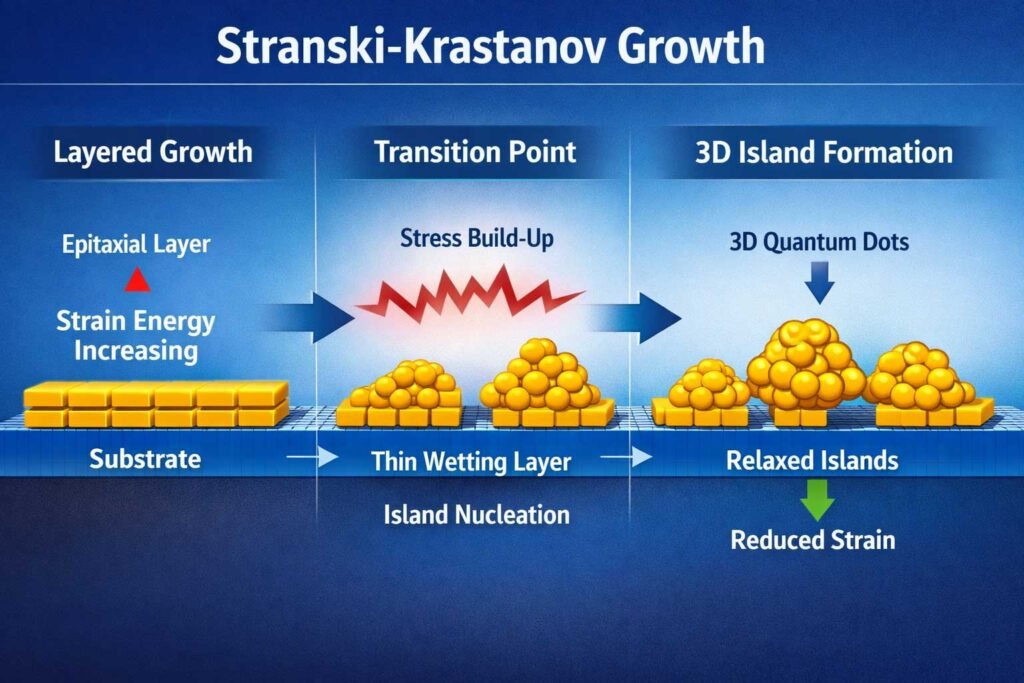
One of Ivan Stranski’s most influential scientific contributions is the Stranski–Krastanov growth model, developed together with Lyubomir Krastanov.
This model describes a fundamental process in materials science that explains how thin films grow on surfaces—and why that growth suddenly changes behavior.
What does it describe?
The Stranski–Krastanov model explains a two-stage growth process:
- Layer-by-layer growth (2D phase)
A material initially spreads evenly across a surface, forming smooth, uniform atomic layers. - Transition to island growth (3D phase)
After reaching a critical thickness, the system becomes unstable, and the material begins forming three-dimensional islands on top of the layers.
👉 This transition is one of the most important phenomena in crystal growth.
Why does this happen?
The shift from flat layers to 3D islands is driven by competing physical forces:
- Surface energy differences
The system seeks to minimize total energy, which changes as layers accumulate - Lattice mismatch between materials
Differences in atomic spacing create strain between the film and the substrate - Mechanical stress buildup
As more layers form, internal stress increases until the structure relaxes by forming islands
Together, these factors make continued flat growth energetically unfavorable, triggering the transition.
Why it matters today
The Stranski–Krastanov growth model is essential in modern technology because it explains how to control structures at the nanoscale.
It plays a critical role in:
- Quantum dot formation – used in displays, lasers, and medical imaging
- Semiconductor fabrication – the foundation of computer chips and electronics
- Microelectronics – enabling smaller, faster, and more efficient devices
- Nanostructure design – controlling material properties at the atomic level
Engineers and scientists actively use this model to design materials with specific electrical, optical, and mechanical properties.
Lasting impact on technology
What makes this discovery extraordinary is its longevity.
Decades after it was first proposed, the Stranski–Krastanov model remains:
- A standard concept in physics and materials science
- A practical tool in advanced manufacturing
- A key principle behind emerging technologies
👉 Without this discovery, many modern technologies—especially in electronics and nanotechnology—would not exist in their current form
It is a perfect example of how deep theoretical insight can lead to transformative real-world applications.
The Kossel–Stranski Model
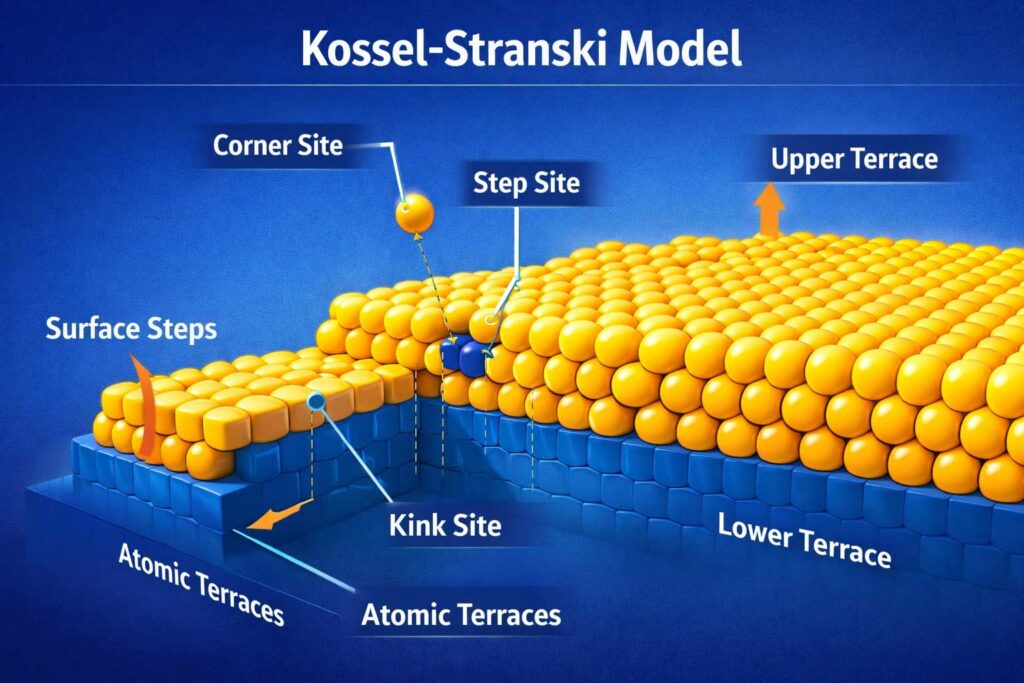
Another major contribution of Ivan Stranski is the Kossel–Stranski model, developed in collaboration with Walther Kossel.
This model provides a fundamental explanation of how crystals grow at the atomic level, transforming what was once a poorly understood process into a structured and predictable phenomenon.
How the model works
The Kossel–Stranski model describes crystal growth as a layer-by-layer process, where atoms attach to specific sites on a crystal surface.
It focuses on three key structural features:
- Step edges – boundaries where a new atomic layer begins to form
- Atomic terraces – flat regions between steps
- Kink sites – highly reactive points where atoms attach most easily
These features determine how atoms move, attach, and stabilize during crystal formation.
A structured view of crystal growth
Before this model, crystal growth was often seen as a somewhat random accumulation of atoms.
The Kossel–Stranski model introduced a powerful new idea:
👉 Crystal growth follows organized, energy-driven pathways at the atomic level
Atoms do not attach randomly. Instead, they:
- Prefer energetically favorable sites
- Build stable configurations step by step
- Form ordered layers over time
This insight was crucial for turning crystal growth into a predictive scientific discipline.
Why it matters today
The Kossel–Stranski model remains one of the core foundations of surface science and materials engineering.
It is still used to understand and design:
- Thin films and coatings
- Semiconductor surfaces
- Nanostructures and quantum materials
- Catalytic surfaces in chemistry
Modern technologies rely heavily on controlling atomic-level growth, and this model provides the conceptual framework for doing so.
Lasting scientific impact
Together with Stranski’s other work, this model helped establish the modern understanding of crystal formation.
It is now:
- Taught in physics and chemistry worldwide
- Applied in advanced research laboratories
- Embedded in the foundations of nanotechnology
👉 What once seemed invisible—the behavior of atoms on surfaces—became measurable, predictable, and controllable
And that transformation is a direct result of Stranski’s work.
International Career and Scientific Influence

Ivan Stranski’s career extended far beyond Bulgaria, placing him among a rare group of scientists who operated at the center of international scientific development in 20th-century Europe.
He collaborated with some of the most influential researchers of his time, including Max Volmer, a leading figure in electrochemistry and surface science. These collaborations helped position Stranski within the forefront of modern physical chemistry.
A truly international scientific path
Stranski’s academic journey took him across several major scientific hubs:
- Germany (Berlin) – the epicenter of European scientific research
- Soviet Union (Ural Institute, Sverdlovsk) – an emerging center for physics and materials science
- Breslau (now Wrocław, Poland) – an important academic and research city at the time
This international exposure allowed him to:
- Exchange ideas with top scientists
- Access advanced laboratories
- Contribute to global scientific discourse
It also strengthened his ability to merge different scientific traditions into a unified theoretical approach.
Leadership at the Technical University of Berlin
At the prestigious Technical University of Berlin, Stranski reached the peak of his academic leadership.
He served as:
- Professor of Physical Chemistry
- Dean of Faculty
- Rector (1951–1953)
These roles placed him at the center of rebuilding and guiding one of Germany’s most important technical universities during the post-war period.
His leadership was not purely administrative—he actively shaped:
- Academic programs
- Research directions
- Institutional recovery after World War II
Work at the Fritz Haber Institute
Later in his career, Stranski worked at the Fritz Haber Institute, one of the world’s most prestigious research centers in physical chemistry and surface science.
There, he continued advancing research in:
- Crystal growth
- Surface phenomena
- Phase transitions
The institute itself has been home to Nobel Prize-winning research, placing Stranski among the top scientific minds working in Europe at the time.
A bridge between scientific worlds
Stranski’s international career made him more than just a researcher—he became a bridge between scientific cultures.
He connected:
- Bulgarian academic development
- German scientific excellence
- Broader European research networks
This unique position allowed his ideas to spread widely and ensured that his work had a lasting global impact.
World War II and Scientific Leadership
World War II was a decisive turning point in Ivan Stranski’s life and career.
As Europe descended into chaos, the scientific world was deeply affected. Universities were damaged or destroyed, research programs were halted, and many scientists were displaced or forced into exile.
During this period:
- Academic institutions across Europe collapsed
- Scientific collaboration was severely disrupted
- Laboratories and research facilities were heavily damaged
Amid this instability, Stranski chose to remain in Berlin—a city at the center of both scientific excellence and wartime destruction.
Leadership in a time of crisis
Rather than withdraw from academic life, Stranski took on a crucial role in preserving and rebuilding scientific institutions.
He became actively involved in:
👉 Reconstructing academic structures after the war
👉 Supporting the continuation of scientific education
👉 Stabilizing research environments in a fractured system
Despite overwhelming challenges, including:
- Allied bombings that devastated Berlin
- Institutional disarray and lack of resources
- Political uncertainty in post-war Germany
Stranski remained committed to science and education.
Reopening scientific life
One of his most significant contributions during this time was helping reopen the Technical University of Berlin.
In 1945, despite severe destruction, the university became one of the first institutions to resume academic activity. Stranski played a key role in:
- Reorganizing departments
- Restoring teaching programs
- Supporting students returning after the war
This effort was critical in ensuring that scientific knowledge and education did not disappear during one of the darkest periods in modern history.
A builder, not just a scientist
Stranski’s actions during and after World War II reveal another dimension of his legacy.
He was not only a theorist and researcher—he was also:
- An institutional leader
- A protector of scientific continuity
- A key figure in rebuilding European academia
At a time when many systems collapsed, Stranski helped ensure that science endured.
Lasting significance
His work during this period demonstrates an essential truth:
👉 Science depends not only on discovery, but on those who preserve and rebuild it
By helping restore academic life in post-war Berlin, Ivan Stranski contributed not just to chemistry—but to the survival of scientific culture itself.
Political Exile and Return to Bulgaria

After the Bulgarian coup d’état of 1944, Ivan Stranski’s life and career entered one of their most difficult phases.
The political transformation in Bulgaria brought sweeping changes across all institutions, including universities and scientific organizations. Like many intellectuals associated with the pre-war academic system, Stranski became a target of suspicion under the new communist government.
He was:
- Accused of connections to the previous regime
- Removed from the academic position he had built
- Gradually marginalized within Bulgarian scientific life
This marked a dramatic shift for a man who had been instrumental in creating the very foundation of physical chemistry in Bulgaria.
Years in exile
Despite these setbacks, Stranski did not disappear from science.
Instead, he continued his work abroad—primarily in Germany—where his reputation remained strong. At institutions such as the Technical University of Berlin and the Fritz Haber Institute, he remained an active and respected researcher.
During this period:
- He continued publishing and teaching
- His theories gained wider international recognition
- His influence in crystal growth research expanded
Ironically, while his homeland distanced itself from him, the global scientific community continued to embrace his work.
Delayed recognition
It was not until the 1960s that Bulgaria began to reassess Stranski’s legacy.
He was:
- Reinstated as a member of the Bulgarian Academy of Sciences
- Officially acknowledged once again as a leading Bulgarian scientist
This late recognition reflects a broader pattern in history, where political systems eventually give way to the enduring value of scientific achievement.
Return to Bulgaria
In 1967, after decades of political exclusion, Ivan Stranski returned to Bulgaria.
His return symbolized more than a personal homecoming—it represented a reconciliation between a nation and one of its greatest scientific minds.
Although much time had passed, his contributions could no longer be overlooked. By the time of his return, his work had already become firmly embedded in global science.
A legacy beyond politics
Stranski’s experience highlights a powerful reality:
👉 Scientific truth is not defined by political systems
Even when institutions fail to recognize it, meaningful scientific work continues to influence the world.
His life stands as a testament to resilience—showing that intellectual legacy can endure long after political circumstances have changed.
Honors and Recognition
Ivan Stranski’s scientific achievements earned him widespread international recognition, placing him among the most respected physical chemists of the 20th century. His work transcended national boundaries and was acknowledged by leading scientific institutions across Europe and the United States.
Membership in prestigious academies
Throughout his career, Stranski was elected to some of the most influential scientific organizations in the world—an honor reserved only for researchers who have made lasting contributions to global science.
He was a member of:
- Göttingen Academy of Sciences – one of Europe’s oldest and most prestigious scholarly institutions
- Bavarian Academy of Sciences and Humanities – a leading center for interdisciplinary research
- New York Academy of Sciences – a globally recognized scientific society connecting international researchers
- Leopoldina – Germany’s national academy and one of the oldest scientific academies in the world
These memberships highlight the global impact of Stranski’s work, particularly in crystal growth theory and surface science.
Institutions named in his honor
Stranski’s influence is permanently embedded in scientific infrastructure through institutions that carry his name.
Among them:
- The Stranski Laboratory for Physical and Theoretical Chemistry at Technical University of Berlin
- The I. N. Stranski Institute of Metallurgy in Germany
These institutions continue to advance research in materials science, physical chemistry, and surface physics, extending Stranski’s legacy into modern scientific innovation.
Recognition in mineralogy
Stranski’s contributions were also honored in the field of mineralogy with the naming of the mineral stranskiite.
This rare mineral serves as a lasting tribute to his work in crystal structures and growth processes—fields that directly relate to mineral formation and atomic arrangement.
National recognition in Bulgaria

Although political circumstances temporarily distanced him from his homeland, Stranski was ultimately recognized as one of Bulgaria’s most important scientists.
Today, his legacy is preserved through:
- A street named after him in Sofia
- His reinstatement as a member of the Bulgarian Academy of Sciences
- Continued recognition in Bulgarian scientific and academic history
Why this recognition matters
Ivan Stranski’s honors are not merely symbolic—they reflect the enduring relevance of his work.
His theories continue to influence:
- Nanotechnology
- Semiconductor engineering
- Surface chemistry
- Advanced materials science
Few scientists achieve recognition across multiple disciplines and multiple countries.
Stranski did.
And that is why his name remains firmly embedded in the foundations of modern science.
Scientific Legacy and Modern Impact

Ivan Stranski’s work continues to influence modern science in powerful ways.
His theories are used in:
- Semiconductor chips
- Nanotechnology
- Solar energy materials
- Quantum computing structures
- Surface engineering
In particular, Stranski–Krastanov growth is essential in:
👉 Quantum dot lasers
👉 Advanced microelectronics
👉 High-performance computing materials
Few scientists achieve this level of lasting relevance.
The rise of Bulgarian science onto the world stage
At a time when Bulgaria was still developing its scientific identity, Stranski proved that world-class research could emerge from anywhere. His work placed Bulgarian science alongside leading European traditions and demonstrated that intellectual excellence is not limited by geography.
Today, he stands as a symbol of Bulgaria’s contribution to global science—an example that continues to inspire new generations of researchers.
The power of theoretical science to shape technology
Stranski’s work was deeply theoretical. He studied how atoms behave, how surfaces evolve, and how crystals form—topics that may have seemed abstract at the time.
Yet today, those same ideas are essential in:
- Microchips and processors
- Nanotechnology and advanced materials
- Renewable energy systems
- Quantum computing
His legacy shows that theoretical science is not separate from reality—it builds the foundation of future technologies.
The resilience of science despite political upheaval
Stranski lived through one of the most turbulent periods in modern history:
- Two World Wars
- Political regime changes
- Academic displacement and exile
Despite these challenges, his work endured. Even when he was removed from his position in Bulgaria, his ideas continued to influence global science.
This highlights an important truth:
👉 Scientific knowledge survives beyond political systems
Science outlives politics
Stranski’s life is a clear example that political environments may change, but scientific contributions remain.
While governments rise and fall, discoveries continue to be studied, applied, and expanded. His theories are still used today in cutting-edge research—decades after the political struggles that affected his career.
👉 Science outlives politics
One mind can shape entire industries
Few scientists leave a legacy that extends into multiple industries.
Stranski did.
His work directly impacts:
- Semiconductor manufacturing
- Nanotechnology engineering
- Quantum dot applications
- Surface and materials science
These are not niche fields—they are the backbone of modern technology.
👉 A single mind can influence entire industries for generations
FAQ About Ivan Stranski
Who was Ivan Stranski?
Ivan Stranski was a Bulgarian physical chemist and one of the pioneers of modern crystal growth theory. He is considered the founder of the Bulgarian school of physical chemistry and played a key role in establishing the field as a rigorous scientific discipline. Through both his research and teaching, he helped connect Bulgarian science with leading European institutions and influenced generations of scientists.
What is Stranski–Krastanov growth?
Stranski–Krastanov growth is a fundamental model in materials science that describes how thin films form on surfaces. Initially, atoms deposit in smooth, uniform layers (layer-by-layer growth). After reaching a critical thickness, strain builds up, causing the material to transition into three-dimensional island structures.
This mechanism is essential for understanding quantum dots, semiconductor fabrication, and nanotechnology, and it remains widely used in modern physics and engineering.
Why is Ivan Stranski important?
Ivan Stranski is important because he helped lay the theoretical foundations of modern materials science and surface chemistry. His work explains how crystals grow at the atomic level, which is critical for technologies such as:
- Semiconductor chips
- Nanomaterials
- Solar cells
- Quantum computing components
In addition, he built the academic framework for physical chemistry in Bulgaria, making him both a scientific pioneer and an institutional leader.
Where did Ivan Stranski work?
Ivan Stranski is not just a historical figure remembered in textbooks—he is a scientist whose ideas continue to shape the modern world in profound and practical ways.
He represents several powerful themes that remain highly relevant today.
Final perspective
Ivan Stranski’s story is not only about scientific discovery—it is about impact.
It is about how one individual, working with ideas at the atomic level, helped shape technologies that define the modern world.
And that is why he still matters today.
👉 Learn more about other innovators like
- Stamen Grigorov and Lactobacillus bulgaricus
- Georgi Nadjakov and the photoelectret effect
- Assen Jordanoff and early aviation engineering
References
Sources include scientific journals, university archives, and authoritative academic institutions to ensure accuracy and credibility.
- Lacmann, R. (1981). “Iwan N. Stranski (2.1.1897–19.6.1979).” Zeitschrift für Kristallographie, 156(1–4), 167–175.
https://doi.org/10.1524/zkri.1981.156.14.167 - Gutzow, Ivan S. (1997). “In Memoriam: Prof. Dr. Ivan Stranski (1897–1979): one of the Founders of the Present-day Theory of Crystal Forms, Crystal Nucleation and Growth.” Crystal Research and Technology, 32(6), 753–758.
https://doi.org/10.1002/crat.2170320603 - Sretenova, Nikolina. “Ivan N. Stranski: An Exciting Journey to the Academic Top.” Union of Scientists in Bulgaria.
https://www.uni-sofia.bg - “Iwan Nicolá Stranski (1897 – Sofia – 1979).” History and Foundations of Quantum Physics.
https://www.mpiwg-berlin.mpg.de - Technische Universität Berlin. “The Shoulders on Which We Stand: Iwan N. Stranski (1897–1979).”
https://www.tu.berlin - Knobloch, Eberhard (Ed.). (2004). The Shoulders on Which We Stand: Wegbereiter der Wissenschaft – 125 Jahre Technische Universität Berlin. Springer, pp. 166–168.
https://link.springer.com/book/10.1007/978-3-540-20557-9 - Пенчев, Бойко (2008). “Проф. Иван Странски (1897–1979).” In 120 години Софийски университет ‘Св. Климент Охридски’: Традицията да бъдеш първи, pp. 80–81. София: Университетско издателство.
https://www.uni-sofia.bg - Technische Universität Berlin. “Rektoren und Präsidenten seit 1946.”
https://www.tu.berlin - Handbook of Mineralogy. “Stranskiite.”
https://www.handbookofmineralogy.org - Mindat.org. “Stranskiite Mineral Data.”
https://www.mindat.org/min-3798.html
Photo credit: Sofia university archives Wikimedia


